Avian Influenza
Avian Influenza
Avian influenza (AI) is a highly contagious viral infection which may cause up to 100% mortality in domestic chickens or turkeys. The disease is caused by a virus that belongs to the family Orthomyxoviridae. Influenza viruses have two surface proteins, haemagglutinin and neuraminidase, that determine their subtype and the animal species they infect; there are 16 haemagglutinin and nine neuraminidase types. When AI viruses of two haemagglutinin types, H5 and H7, infect domestic poultry (chickens or turkeys) they often mutate and virulent disease arises in these birds which is called highly pathogenic avian influenza (HPAI). The initial infection that does not cause or causes minimal disease is called low pathogenic avian influenza (LPAI). Wild water birds act as reservoir hosts of influenza viruses, however, these viruses generally do not cause disease in these birds.
Birds susceptible to AI

Avian influenza can cause mass mortality Source: CSIRO
All commercial, domestic and wild bird species are susceptible to infection with AI viruses but disease outbreaks occur more frequently in chickens and turkeys. LPAI viruses are traditionally spread by migratory wild birds. Many species of waterfowl, especially geese, ducks and swans, carry the virus but generally show no signs of disease. The recent HPAI outbreak in eastern Asia has seen the H5N1 virus cause disease and high mortality in ducks, geese, swans and other wild birds.
Avian influenza in Australia
While LPAI viruses are probably ubiquitous throughout the world in wild water birds, outbreaks of HPAI have occurred as sporadic events that were controlled and eradicated until the recent eastern Asia outbreak. The H7 subtype HPAI virus has caused outbreaks of clinical disease in commercial poultry in Australia in Victoria (1976, 1985 and 1992), Queensland (1994) and NSW (1997). All outbreaks were quickly eradicated. Avian influenza as it occurs in Asia as H5N1 subtype virus has not been recorded in Australian poultry flocks.
AUSVETPLAN
The Australian strategy is to eradicate the virulent disease by immediate culling and disposal of infected and in-contact birds to remove the major source of the virus. Additionally, there will be strict quarantine and movement controls to prevent the spread of infection, decontamination to remove and destroy the virus, tracing and surveillance to locate the source of infection, locate other infected premises and determine the extent of the infection and zoning to define infected and disease-free areas. Vaccination may be an option in some circumstances and provisions have been made for emergency vaccine supplies to be available for use in Australia. The current Emergency Animal Disease Response Agreement applies to both HPAI and LPAI outbreaks (see AUSVETPLAN – Avian Influenza). Government and the poultry industries share the costs of eradication under the Emergency Animal Disease Response Agreement, which is administered by Animal Health Australia.
H5N1 virus
The spreading Asia outbreak is unprecedented in recent history. The H5N1 virus was first detected in China in 1996 and the infection spread to other eastern Asian countries in 2003 and evolved to produce severe disease and deaths in domestic and wild water birds, which is an unusual feature for AI viruses. Altogether more than 150 million domestic birds have died or been killed in the attempt to control AI infection in eastern Asia. In mid-2005, the infection spread eastwards to countries bordering the Black Sea with infection occurring in both wild birds and poultry. In early 2006, H5N1 infection spread to Nigeria, India and Egypt in poultry and to many countries in Europe with deaths in wild birds, particularly swans.

Avian influenza causing depression Source: CSIRO
Significance of AI outbreaks
Since 1997 when H5N1 virus was shown to infect humans in Hong Kong, there has been increasing concern about HPAI poultry outbreaks. With more than 170 confirmed human infections and over 90 deaths in Asia, the World Health Organisation (WHO) is concerned about a pandemic of human influenza arising from mutation(s) of the poultry virus or its recombination with a human influenza virus that will enable the virus to infect and spread between people easily.
Recently the World Organisation for Animal Health (OIE) has redefined avian influenza infection as both HPAI and LPAI viruses of the H5 and H7 subtypes and in so doing indicated there is a need to control both LPAI and HPAI infections in domestic poultry and other domestic birds. It is not practical to control AI infections in wild bird populations and it is recommended that action not be taken to control infection in these species when outbreaks occur.
Spread of infection
Direct or indirect contact (likely through drinking water) with migratory waterfowl is the most likely source of infection for domestic poultry. Once established in domestic poultry, infection can also spread through contact with contaminated equipment or humans. Transmission through the egg is uncommon, although contamination of the shell does occur. Avian influenza virus is highly concentrated in the manure and in nasal and eye discharges.
Avian influenza in poultry and impact on the poultry industries
The clinical signs of infection with HPAI virus are variable and can be affected by the existence of other diseases, the age of the birds, the environment and the severity of the virus itself. In very severe forms the disease appears suddenly and birds die quickly. Some may appear depressed, egg production falls and softshelled eggs are produced. There may be profuse watery diarrhoea, combs and wattles may become blue and respiration may be laboured. With less virulent forms of HPAI, the clinical signs may include decreased egg production, depression, respiratory signs suggestive of a cold, swelling of the face, possibly some nervous signs and diarrhoea. With LPAI, there may be no clinical signs seen following infection or mild signs relating to the respiratory, alimentary or reproductive systems may be seen.
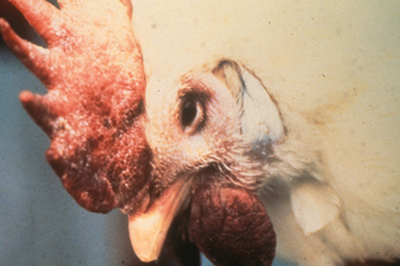
Avian influenza causing swollen head Source: CSIRO
Persistence of AI virus
Environmental conditions have a marked effect on virus survival outside the bird. Avian influenza virus can survive for at least 35 days at 4°C in manure and can be isolated from lake water where waterfowl are present. The virus can survive for up to 23 days if refrigerated and for several days in carcases at ambient temperature. The virus can persist in poultry meat products but is eliminated by cooking.
Prevention of infection
Australia has developed industry plans (called biosecurity plans) to prevent avian influenza viruses gaining entry to commercial poultry flocks. The plans are aimed at limiting possible contact between wild birds and domestic poultry through contaminated water and food supplies and transfer of infection by the mechanical movement of infection on fomites such as on the clothing and footwear of persons and on equipment, containers, vehicles etc. Treatment of surface water by chlorination to inactivate the virus is essential if it is to be supplied to poultry and aviary birds.
Suspicion of AI
If you are in contact with commercial poultry which present with any unusual disease signs, abnormal behaviour or unexplained deaths, or with pet birds with depression, rapid breathing and a sudden rise in mortality, report this to your veterinarian or Department of Primary Industries/Agriculture Officer or alternatively call the Emergency Animal Disease Watch Hotline on 1800 675 888 at once.
Control of AI outbreaks
Australia has very strict quarantine procedures that seek to prevent the entry of live birds and poultry products from countries that have experienced AI outbreaks and increased surveillance is undertaken at airports, seaports and international mail exchanges when AI outbreaks occur overseas. There is nothing that can be done to control the infection in wild birds but the likelihood of migratory birds introducing H5N1 infection to Australian poultry is regarded as highly unlikely given wild birds become rapidly ill or die after becoming infected and the main reservoir hosts (ducks, geese and swans) do not migrate into Asia. The use of AI vaccination will likely be considered in future in the event of an outbreak that is not readily controlled by eradication techniques.
Control of AI outbreaks Avian influenza viruses and human infection
Generally, outbreaks of AI infections in poultry or wild birds have not been associated with cases of human influenza. However cases of human influenza due to H5 and H7 subtype HPAI viruses have been recorded in a few humans associated with recent poultry outbreaks in Europe (2003), eastern Asia (1997–2006) and western Asia (2006).
Human pandemic influenza
Human pandemic influenza occurs irregularly, having occurred in 1919, 1957 and 1968 when strains of influenza virus with a novel haemagglutinin type adapted to humans and spread rapidly around the world. It is feared that another human pandemic will arise from the current AI outbreak in eastern Asia. Illness and death have been associated with outbreaks of HPAI viruses of both H5 and H7 subtype, particularly the H5N1 virus in eastern Asia. The pandemic of human influenza will not occur until there have been mutations in the poultry H5N1 virus or its recombination with a human influenza virus that will allow the resulting virus to easily infect and transmit between humans. To this time, contact with live infected birds or their manure has been necessary for infection to establish in humans and few human cases have infected other humans in contact with them. A pandemic cannot commence without an AI virus acquiring the ability to easily infect and spread between humans. Avian influenza in birds and human pandemic influenza can be expected to be caused by genetically different viruses.
Dangers for humans consuming poultry products
The recognised source of AI infection for humans is direct contact with infected live birds or their manure, so poultry products are relatively safe. Handling raw poultry meat products or eggs and eating cooked poultry products have not been recorded as causing infection. Cooking poultry products in the normal way destroys AI viruses.
Further information
- Original article written by Dr Andrew Turner
- Events are always changing so websites are recommended here for further reading. Two useful Australian websites for monitoring current events with an Australian perspective are: DAFF.gov.au and HEALTH.gov.au
- Public health information is available on the hotline 1800 004 599.
- The Food and Agriculture Organisation of the United Nations (FAO) has provided regular updates on the worldwide situation of Avian influenza occurrence for the last two years. Previous and current updates are available at the following site: FAO.org
- The World Health Organisation (WHO) has a website updating information about the planning for dealing with a pandemic and human infections that have occurred with H5N1 virus. The information can be obtained at: WHO.int
- The International Association for Food Protection (IAFP), in the light of the H5N1 outbreak in eastern Asia, has issued a statement on the safety of consuming poultry products and measures that will inactivate the virus should poultry or eggs be contaminated with AI virus. Information is available at: FoodProtection.org

